Costacurta manufactures wire cloths and meshes for diaphragm cells used to produce caustic soda (chlorine-caustic soda process).
The chlorine-soda process is an electrochemical process that enables the production, through electrolysis, of chlorine gas, caustic soda and hydrogen, starting from a sodium chloride solution (brine). Chlorine and soda can be produced using diaphragm cells, membrane cells or mercury cells.
Do you have a specific request?
Contact us now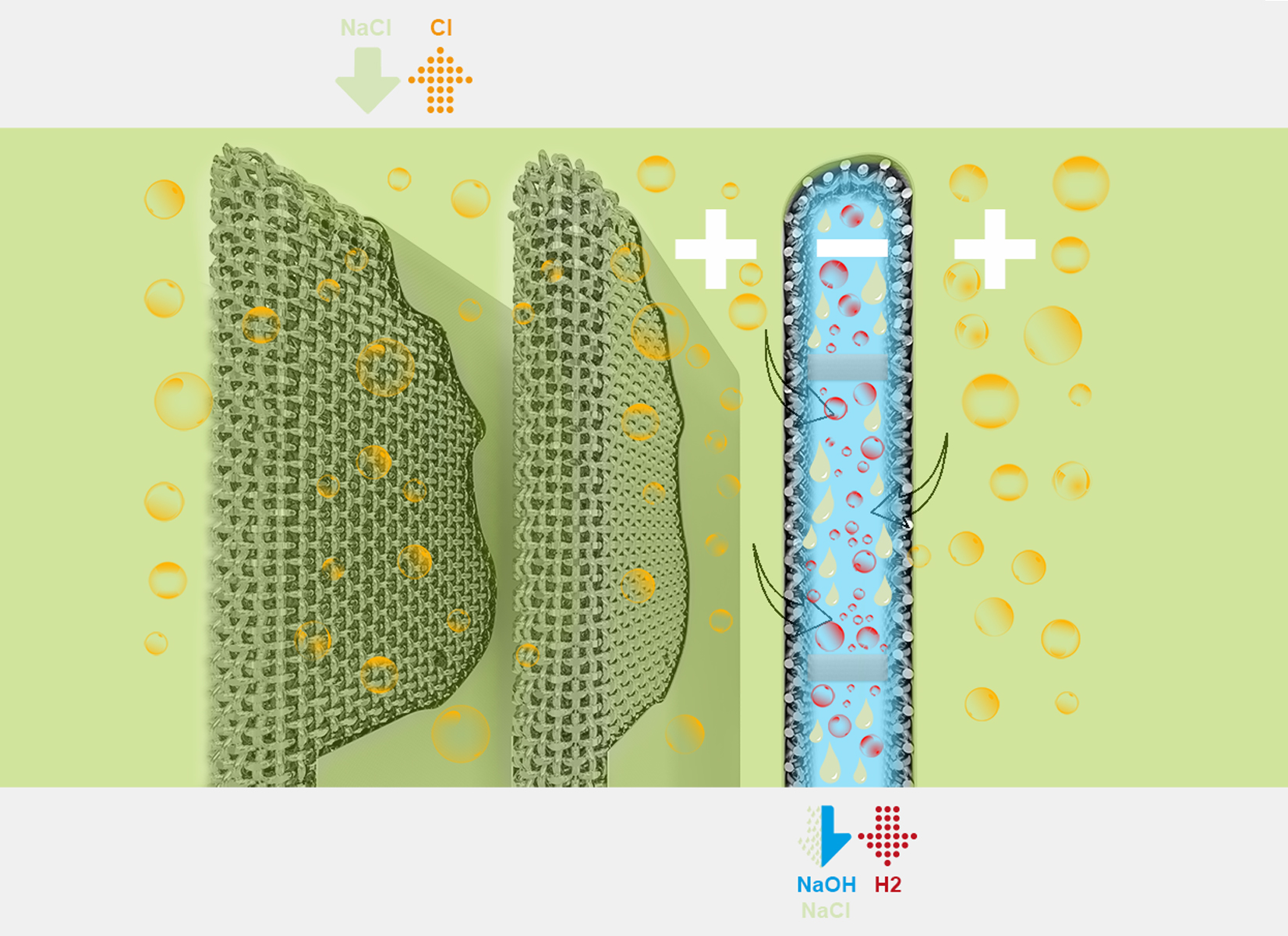
Wire cloths for diaphragm cells used to produce caustic soda
Costacurta’s wire cloths are used in the chlorine-soda process inside diaphragm cells.
During this process, to prevent the chlorine that develops at the anode coming into contact with the catholyte, the anodic compartment is kept separate from the cathodic compartment by an ion-permeable diaphragm that is impermeable to Cl2 molecules.
The sodium chloride solution (anolyte) is placed in the anodic compartment and flows, under hydraulic head, into the cathodic one.
In the first compartment, the Cl ions are oxidised to produce chlorine gas. In the second compartment hydrogen and sodium hydroxide is produced (NaOH, caustic soda).
The cathode units of diaphragm cells (bottom sieves and “cathodic fingers”) are made with a special carbon steel wire cloth, produced by Costacurta.
Costacurta is able to supply custom-made flat panels or shaped and welded individual elements (cathodic fingers), according to the client’s technical specifications and construction drawings.
Distinctive characteristics of the Costacurta metal cloths for diaphragm cells are:
- Weft wires suitably laminated so as to give the metal cloth a relatively flat and uniform surface.
- Constant controlled thickness of the wire cloth
- High quality of the material used and the production process







